The metallic character increases in descending the group. The group is starting from the element that is non-metal and the last elements are non-metals. 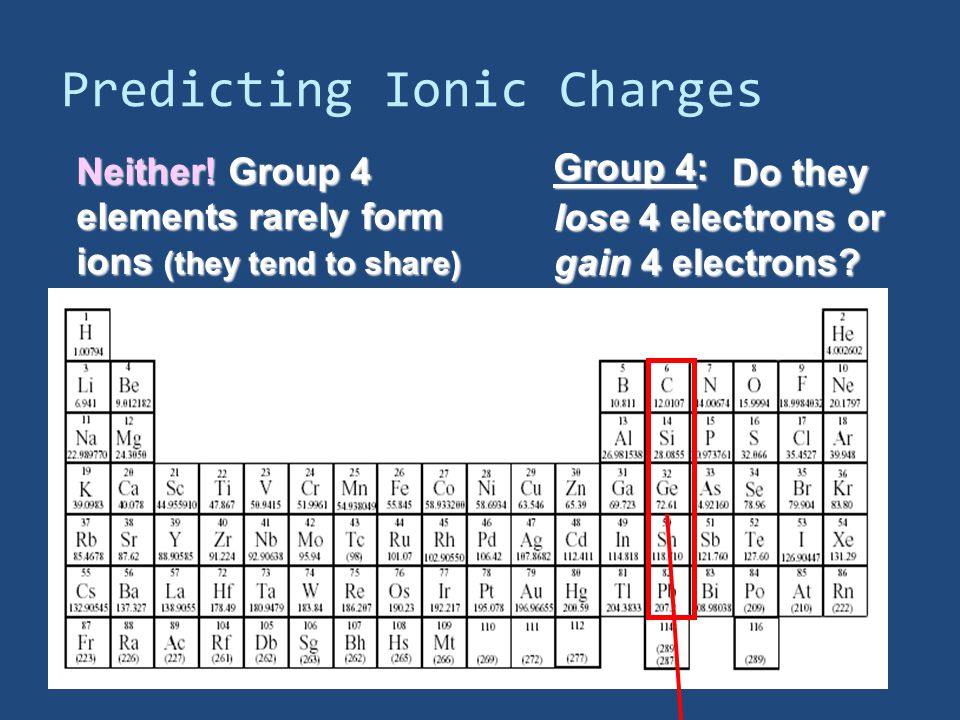
The elements of this group exhibit the covalent oxidation state of +4.Ĭarbon is the first element of this group and is different from the other members due to its high electronegativity, absence of d orbitals and small size. The outer electronic configuration of the elements of this group is ns 2 np 2 whereas n is variable and it varies from 2 to 6. This group consists of carbon, silicon, germanium, tin, and lead.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
